Getting to know the Gut!

A few weeks ago, I had an online chat with Jo-san, one of the founders of Leave a Nest Co., Ltd. The initial aim was to interview him for one of the blog articles to get to know about his journey with Leave a Nest, but we also ended up talking more about his passion for immunology, and specifically, on intestinal microbiota! This gave me an idea on what to cover as my next science communication topic.
Jo-san has always been passionate about science and research. As such, he set up Leave a Nest together with the other founders and has split his time between working at the university as well as developing the ecosystem for science, technology and research through Leave a Nest Japan. He stressed the importance of continuously learning and researching, as well as sharing your findings and passion with those younger than you, because they would be the ones to carry on research in the field in future.
He had also introduced me to two Japanese researchers who would be presenting their works at the 4th Microbiome R&D and Business Collaboration Congress & 3rd Probiotic Congress: Asia to be held in Singapore in March 2018. They are:
- Hiroshi Ohno: Group Director, Laboratory for Intestinal Ecosystem, RIKEN Centre for Integrative Medical Sciences (IMS) Japan
- Jun Kunisawa: Head, Vaccine Materials and Gut Environmental Systems Lab, National Institutes of Biomedical Innovation, Health and Nutrition (NIBIHON), Japan
I thus decided to read about their latest research, to understand what is happening in the field of intestinal microbiome at present. As part of my quest for knowledge, I also stumbled upon other articles, some TED videos, and an initiative called the American Gut Project. I will summarise what I’ve learnt here.
As you might already know, our intestines is a huge organ very compactly fitted into the abdominal region of our bodies. Due to its sheer length, it takes hours for food to pass through it, nutrients to be extracted from it, and for the waste material to be excreted from our body. Due to it’s association to waste, people tend to shy away from talking about it, though recently more emphasis is being placed on understanding our gut because of its association with diseases involving other organs, and general well-being.
There are both “good” and “bad” microbiota living in our intestines, and the balance of the two has important implications on how susceptible we are to diseases. In the last decade, scientists have also been better able to study the microbiota due to the human genome project and advancement in technology, allowing us to study the sequence of human cells, bacteria, etc. Understanding what microbiota are present and how they function has been important in understanding the pathway of how diseases develop and what we can do to prevent or treat them. In some cases, the solution has been as simple as modifying our diets. For example, studies of probiotic bacteria suggest that through producing certain acids, they interact with immune cells in a manner to suppress inflammatory responses, thus having good outcomes in curtailing allergies.
 Due to globalisation, our diets and genetic variation are also changing and consequently, conditions such as Inflammatory Bowel Disease (IBD-Crohn’s and Ulcerative Colitis) are on the rise in Japan and Singapore. IBD is a result of our immune system reacting abnormally, and while the symptoms such as diarrhoea and bleeding ulcers can be treated, there is no cure. I had learnt about these conditions during my studies in Australia, but it interesting to note that this is not just affecting those living in “western countries”, and that through collaboration we could better understand the pathogenesis of these diseases and how to treat them.
Due to globalisation, our diets and genetic variation are also changing and consequently, conditions such as Inflammatory Bowel Disease (IBD-Crohn’s and Ulcerative Colitis) are on the rise in Japan and Singapore. IBD is a result of our immune system reacting abnormally, and while the symptoms such as diarrhoea and bleeding ulcers can be treated, there is no cure. I had learnt about these conditions during my studies in Australia, but it interesting to note that this is not just affecting those living in “western countries”, and that through collaboration we could better understand the pathogenesis of these diseases and how to treat them.
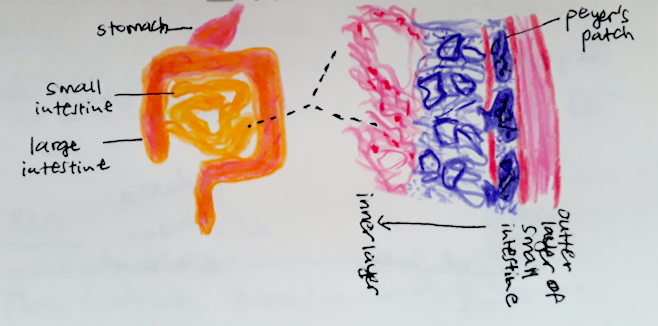
It is very interesting to learn about the array of research out there focusing on the gut and learning how human cells and bacterial cells interact. It seems to be a battle between the two and we need to ensure the immune system doesn’t become overactive and kill off the good bacteria, and at the same time the bad bacteria isn’t strong enough to weaken our immune system and make us sick. In Hiroshi Ohno’s team’s latest published research, they studied Interleukin-22, an immune system molecule and how its level can affect whether bacteria is transported into Peyers Patches, which is a special tissue in our intestines that acts as a surveillance system for our small intestine.
With time, this research could be translated into commercially available products (such as probiotic supplements), that could assist us in maintaining the health of our gut. I hope to learn more about that domain of the science and technology ecosystem, and share with you more in a later post!
Useful Links
- TED video on Rob Knight, involved in Human Microbiome Project: https://www.youtube.com/watch?v=i-icXZ2tMRM
- TED video on Giulia Enders, and her interest in studying the gut: https://www.youtube.com/watch?v=HNMQ_w7hXTA
- The American Gut Project, which allows anyone in the world to learn about the microbiome in their gut: http://americangut.org/
References
- Shinomura, Y. (2016). Trends in Gastrointestinal Diseases in Japan. Digestion. 93, pp 5-6.
- Siah, K.T., Wong, R.K., Chan, Y.H., Ho, K.Y. and Gwee, K.A. (2016). Prevalence of Irritable Bowel Syndrome in Singapore and Its Association with Dietary, Lifestyle and Environmental Factors. Journal of Neurogastroentrology and Motility. 22 (4), pp 670-676.
- Shibata, N., Kunisawa, J., and Kiyono, H. (2017). Dietary and Microbial Metabolites in the Regulation of Host Immunity. Frontiers in Microbiology. 8, 2171.
- Jinnohara, T., Kanaya, T., Hase, K., Sakakibara, S., Kato, T., Tachibana, N., Sasaki, T., Hashimoto, Y., Sato, T., Watarai, H., Kunisawa, J., Shibata, N., Williams, I.R., Kiyono, H., and Ohno, H. (2017). IL-22BP dictates characteristics of Peyer’s Patch follicle-associated epithelium for antigen uptake. Journal of Experimental Medicine. 214 (6), 1607-1618.